Understanding the GLP-1 Receptor Agonist Safety Profile
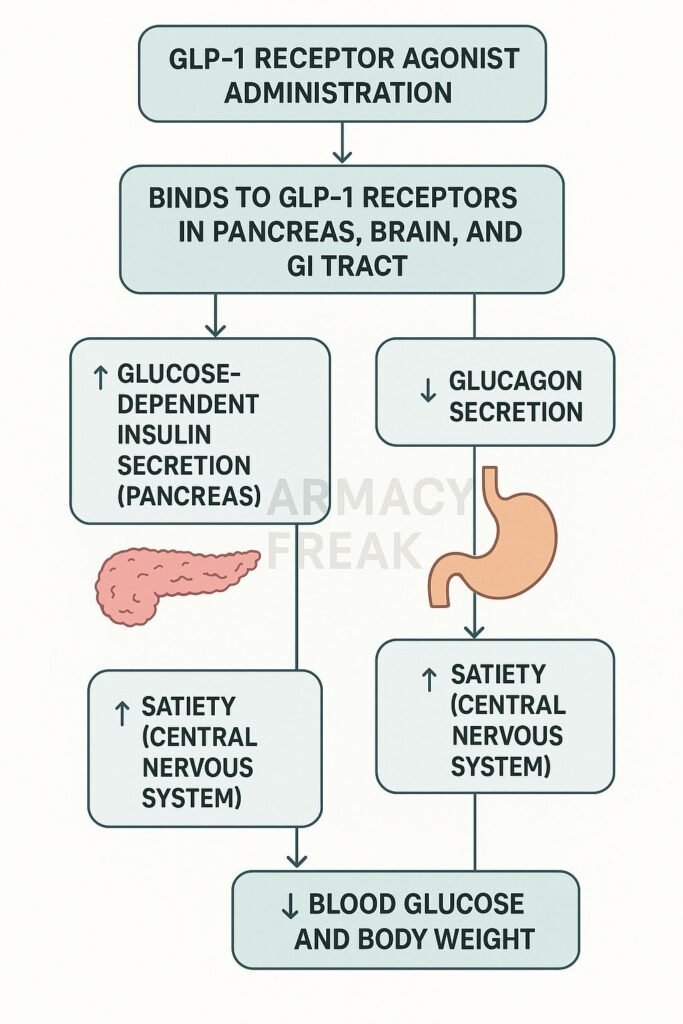
GLP-1 receptor agonists have emerged as a vital option for managing type 2 diabetes and obesity due to their favorable effects on glucose regulation and weight reduction. These medications mimic incretin hormones naturally produced in the intestines, stimulating insulin release when glucose levels rise and slowing gastric emptying. However, like any pharmacological intervention, the use of GLP-1 receptor agonists is not without risks and adverse effects.
Adverse Effects of GLP-1 Receptor Agonists
Common adverse effects of GLP-1 receptor agonists include gastrointestinal (GI) symptoms, such as nausea, vomiting, and diarrhea. Other potential side effects include headaches, fatigue, and injection site reactions. However, a significant number of pancreatitis cases have been reported in association with GLP-1 receptor agonist use.
Risk of Pancreatitis with GLP-1 Receptor Agonists

The risk of pancreatitis associated with GLP-1 receptor agonists is a concern. In the UK alone, over a thousand reports of pancreatitis have been made via the "Yellow Card" scheme since 2007, with 19 deaths reported. This highlights the need for careful monitoring of patients on these medications.
GLP-1 Receptor Agonist Safety Profile in Terms of Kidney Function
Most kidney-related side effects are mild and temporary. GLP-1 receptor agonists work differently than traditional diabetes medications, offering dual metabolic and renal benefits. These medications can help protect kidney health in patients with type 2 diabetes.